

“The Core Force in the Upstream of the Cell Therapy Industry”
Founded in 2018, Weiye Biotech specializes in the research, development, and production of serum-free cell culture media. The company has taken the lead in completing NMPA (National Medical Products Administration) filings for multiple products.
We operate under a strict GMP quality system to ensure product safety and consistency, and have established collaborations with leading research and clinical institutions.
Amid the rapid growth of the cell therapy industry, Weiye Biotech is committed to delivering innovative, compliant, and high-quality traceable raw materials—accelerating localization of supply chains and clinical translation.
“Advancing the frontiers of cell therapy through independent R&D”
Weiye Biotech adheres to a technology-driven development philosophy, continuously increasing investment in the independent R&D and clinical translation of cell culture media.
Our R&D team brings together leading scientists from China and abroad, with 27 core patents in place and a comprehensive technology system covering formulation design, process optimization, and quality control. Each serum-free formulation requires more than five years of development, creating a high-barrier technological moat.
By adhering to rigorous GMP standards and a clinical demand-oriented approach, we are advancing the large-scale application of cell culture media in stem cell and immune cell therapies.
“High-Quality Culture Media to Advance Cell Therapy”
Weiye Biotech is committed to driving continuous innovation in cell culture media through independent R&D and strict regulatory compliance, providing safe and stable core raw materials for stem cell and immune cell therapies. We believe that high-quality culture media serve as the bridge between life sciences and clinical applications. Looking ahead, the company will continue to push technological boundaries, expand its product portfolio and application scenarios, and ensure that cutting-edge medical advances truly benefit society and safeguard patients’ health and lives.
Leveraging a rigorous GMP quality system and proprietary intellectual property, Weiye Biotech has established 27 core patents. Our R&D team, led by top experts in stem cell and immunology research, has built a full-spectrum innovation capability that spans from basic research to clinical translation. Today, the company is among the few domestic suppliers capable of delivering clinical-grade, compliant culture media, and has established long-term collaborations with multiple national research and clinical platforms.
In the future, Weiye Biotech will continue to expand its product pipeline, covering areas such as exosomes, organoids, iPSCs, and dendritic cells. By providing more efficient and safer core raw materials for the rapidly growing cell therapy market, we aim to accelerate the transformation of life science innovations into real-world clinical benefits, safeguarding human health.
With proprietary core patents and a top-tier team, Weiye Biotech has completed regulatory filings for 6 serum-free culture media products and continues to expand its cutting-edge pipeline, building innovation capabilities that span from research to clinical applications.
Serum-free formulations with no animal-derived components, fully compliant with GMP and NMPA filing standards, delivering industry-leading cell expansion efficiency and batch-to-batch consistency.
Backed by a GMP-based quality control system and an independent testing platform, our products demonstrate purity and viability levels far exceeding national standards, ensuring safety, stability, and full traceability.
Our products
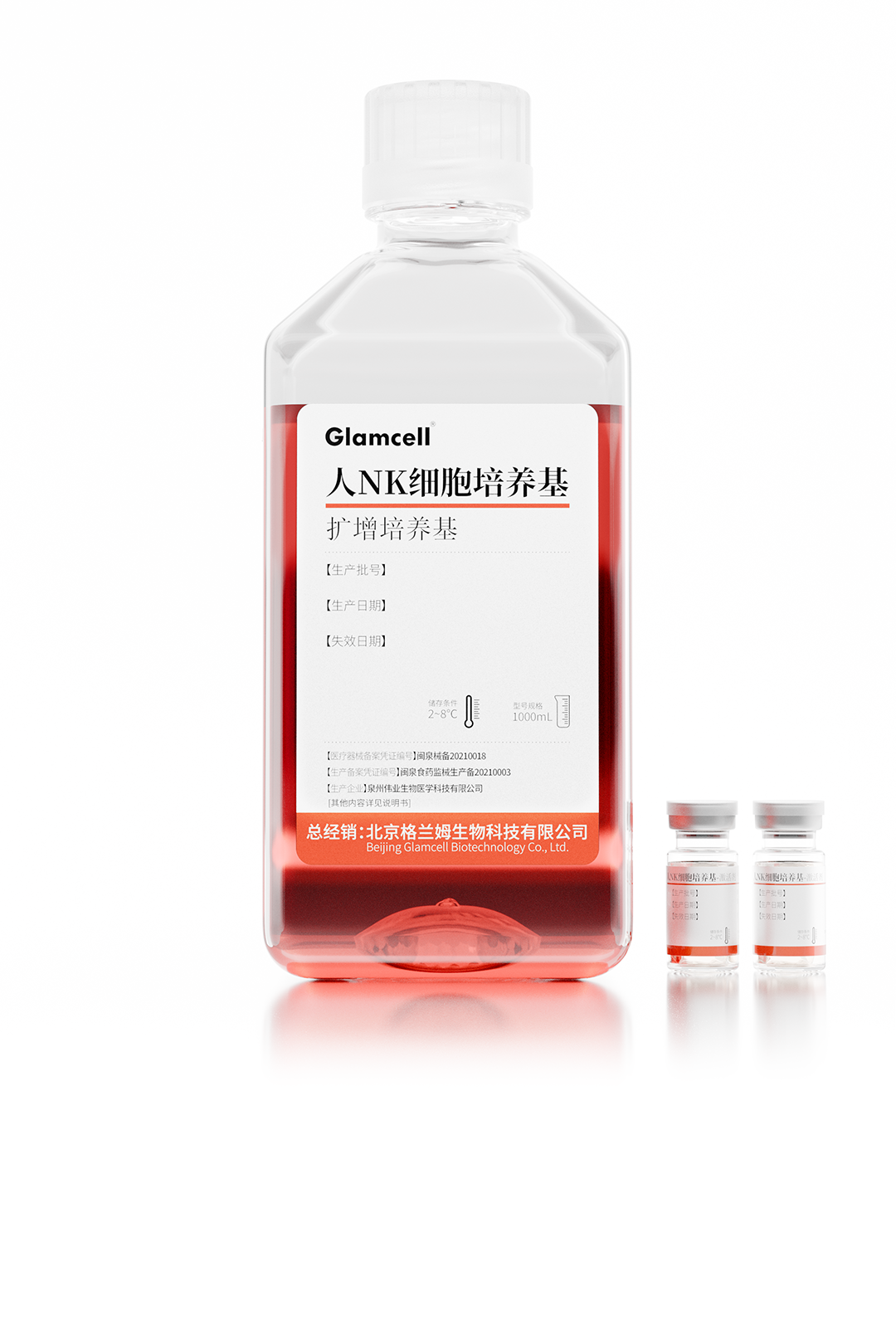
Human NK Cell Culture Medium

Human CIK Cell Culture Medium

Human CINK Cell Culture Medium

γδ T Cell Culture Medium
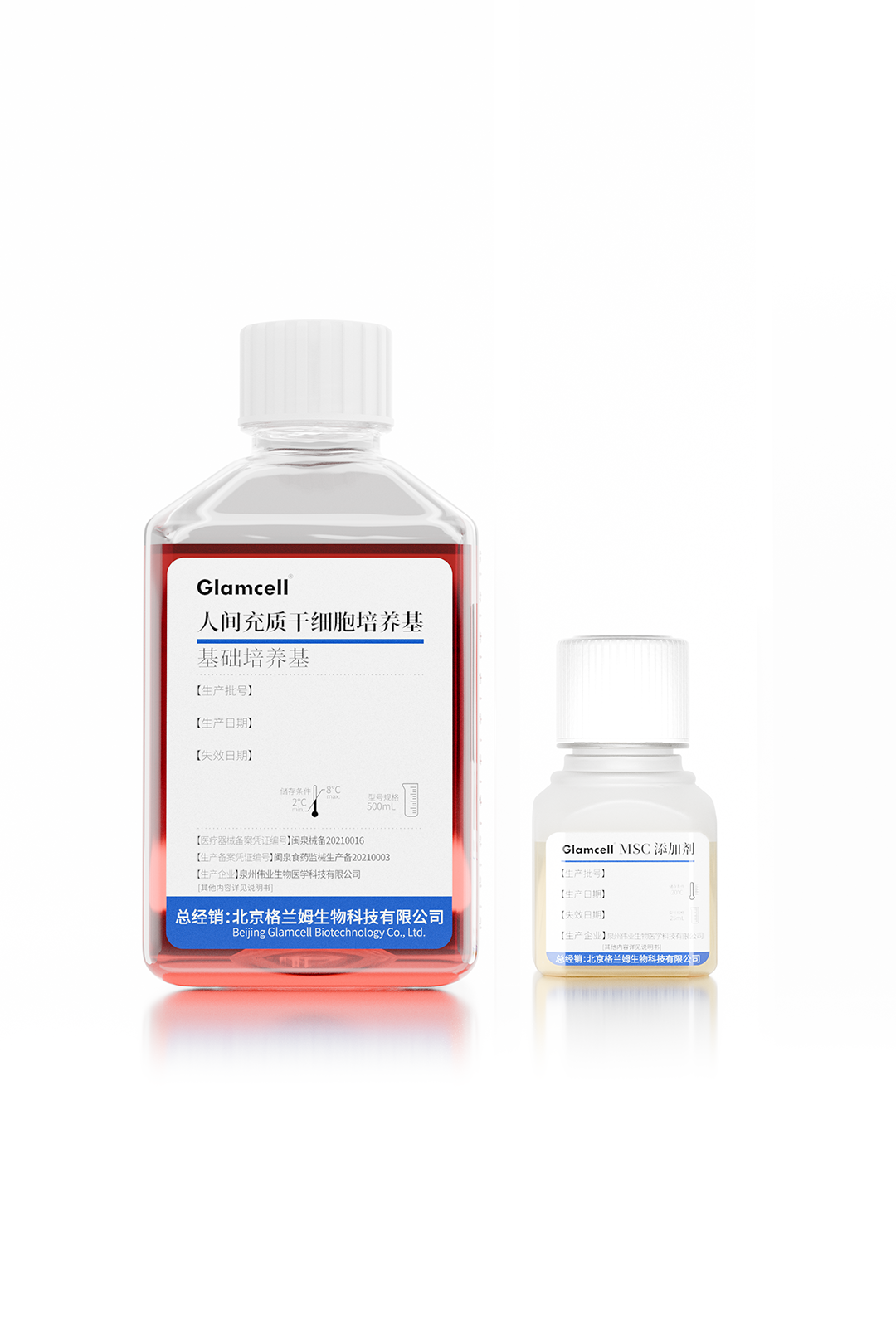
Human MSC Culture Medium
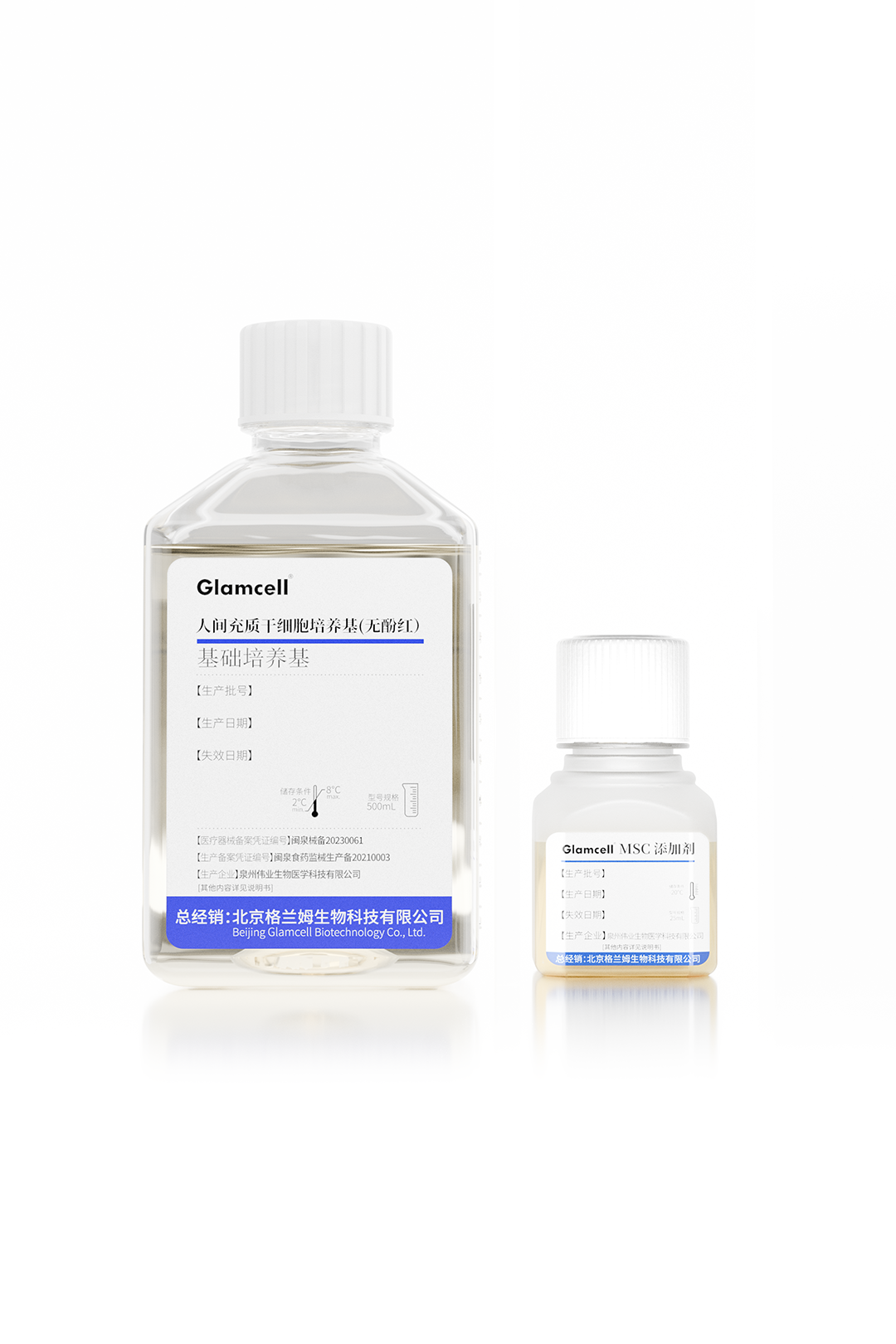
Human MSC Culture Medium
(Phenol Red-Free)
Weiye Biotech has successfully launched six serum-free cell culture media products, all filed with the NMPA, covering key areas including mesenchymal stem cells (MSC), NK cells, CIK, CINK, and γ/δ T cells.
With fully chemically defined formulations and a rigorous GMP quality system, our products lead the domestic market in cell expansion efficiency, batch-to-batch consistency, and regulatory compliance. As a critical upstream raw material for cell therapy, Weiye’s media not only help downstream partners reduce costs by more than 30%, but also play a pivotal role in strengthening local supply chains within the rapidly growing stem cell and immune cell therapy markets.